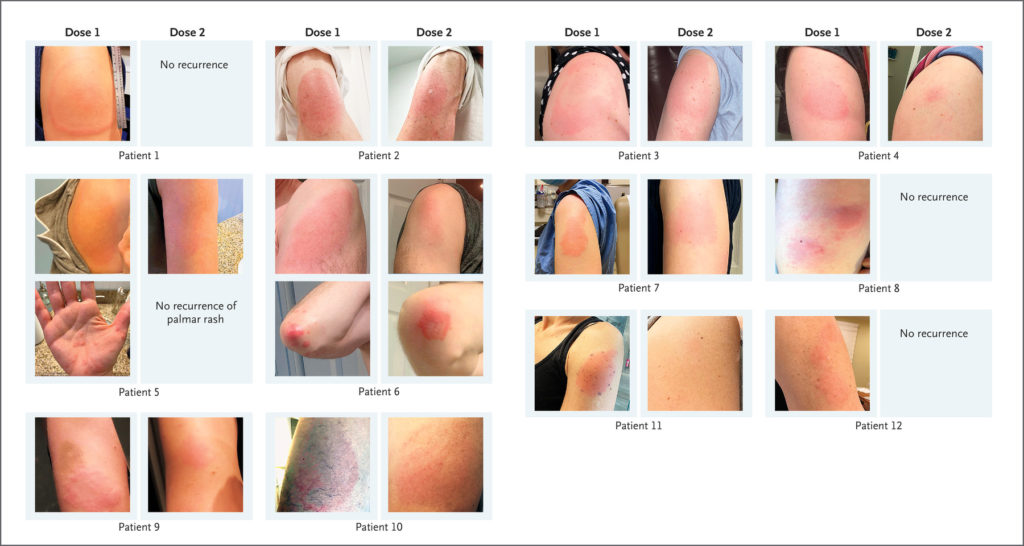
Clinicians should be aware of delayed injection-site reactions that may occur in some recipients of Moderna’s COVID-19 vaccine and which may be confused with skin infection, say a group of physician-researchers from Massachusetts General Hospital.
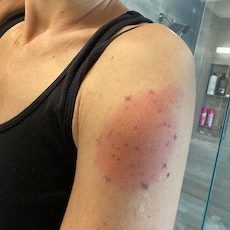
In a letter to the editor in The New England Journal of Medicine, the authors provided their own clinical observations, including images. In a series of 12 patients, symptoms had a median onset at day eight after vaccination, and lasted four to 11 days. Photographs show the varied size and severity of the reactions after both the first vaccination and the second dose (six of the 12 patients experienced a second dose recurrence).
Most patients were treated with ice and antihistamines, but some required corticosteroids, and one was erroneously treated with antibiotics.
These skin reactions were described in the Moderna vaccine’s original trial data, the physician-researchers said. Phase 3 trials found delayed “skin hypersensitivity” in a small number of the more than 30,000 trial participants. The skin reactions were large, red, sometimes raised, itchy or painful. They typically resolved within a week, but were never fully characterized in the initial research, the Massachusetts General physicians said.
In addition, any links between reactions after the first and second doses were not provided, they wrote. “These reactions have not been consistently recognized, guidance regarding the second dose of vaccine has varied, and many patients have unnecessarily received antibiotic agents,” they cautioned.

“Delayed cutaneous hypersensitivity could be confused — by clinicians and patients alike — with a skin infection,” said co-author Erica Shenoy, M.D., Ph.D., associate chief of the Massachusetts General Hospital Infection Control Unit. “These types of reactions, however, are not infectious and thus should not be treated with antibiotics.”
Although the authors emphasize that the reactions should not discourage people from getting vaccinated, clinicians should be prepared to recognize them and guide patients on treatment options and about subsequent completion of a second vaccine dose, they said.
“Whether you’ve experienced a rash at the injection site right away or this delayed skin reaction, neither condition should prevent you from getting the second dose of the vaccine,” said lead author Kimberly Blumenthal, M.D., MSc, co-director of the clinical epidemiology program in the division of rheumatology, allergy and immunology.