
Superbugs are gleaning a lot of attention from the news media lately — justifiably so! They threaten ill and compromised patients, particularly those in long-term care settings. They are resistant to most antibiotics and can spread widely within hospitals and long-term care facilities.
“These are nightmare bacteria that present a triple threat,” said Thomas Frieden, director of the Centers for Disease Control and Prevention. “They’re resistant to nearly all antibiotics. They have high mortality rates, killing half of people with serious infections. And they can spread their resistance to other bacteria.”[i]
The goal is to diagnose such organisms early, treat them appropriately and prevent their spread to other patients. The proper antibiotic selection is key to treatment and preventing resistance. In addition, proper treatment prevents the antibiotic resistant gene from transfer to other bacterial strains.
There are many acronyms for specific organisms and their primary antibiotic resistance, such as CRE, MRSA, ESBL and VRE, but they are all grouped into the lay term of “superbugs.”
CRE
The newest antibiotic resistant organism is Enterobacteraciae–one of the most common bacterial families that cause infection. E. coli is a commonly found example of Enterobacteraciae. From 2001 to 2011, the percent of Enterobacteraciae organisms that are resistant to the most powerful antibiotics (carbapenems) has risen from 1.2% to 4.2%.[ii] Such organisms are called CRE or carbapenem-resistant Enterobacteriaceae.
Healthy people don’t usually get CRE infections. CRE infections are usually secondary to some other disease or malady. Patients whose care requires devices like ventilators (breathing machines), urinary (bladder) catheters, or intravenous (vein) catheters, and patients who are taking long courses of certain antibiotics are most at risk for CRE infections. Obviously this includes both hospitalized and long-term care patients. The CDC article on CRE can be seen here.
MRSA
Methicillin-resistant Staphylococcus aureus (MRSA) infection is caused by a strain of staph bacteria that has become resistant to the antibiotics commonly used to treat ordinary staph infections. MRSA can colonize the nasal passages and can easily be passed from individual to individual. Methicillin is a modern day derivative of penicillin.
Most MRSA infections occur in people who have been in hospitals or other healthcare settings, such as long-term care and dialysis centers. Such infections are known as healthcare-associated MRSA (HA-MRSA). HA-MRSA infections typically are associated with invasive procedures or devices, such as surgeries, intravenous tubing or artificial joints.
Another type of MRSA infection has occurred in the wider community — among healthy people. This form, called community-associated MRSA (CA-MRSA), often begins as a painful skin boil. It’s spread by skin-to-skin contact. At-risk populations include groups such as high school wrestlers, football players, child care workers and people who live in crowded conditions.
Identification of MRSA is often a two-step process. First the organism is isolated and identified as Staph aureus, then subsequently tested for resistance to methicillin. However, some laboratories, such as ours, use a special chromogenic media that allows these two steps to be combined into one, thus shortening the resistant identification of the organism is as little as 24 hours
ESBL
Extended Spectrum Beta-lactamase resistance is prevalent in E.coli, Proteus and Klebsiella—common organisms found in most healthy individuals digestive tract. Extended-spectrum beta-lactamases (EBSL) are enzymes made by certain kinds of bacteria that can break down several types of antibiotics making the infection harder to treat. ESBL is mainly spread among people in hospitals and long-term care facilities. Infections due to ESBL usually affect the urinary tract and intestine, but can also infect wounds and blood. Sometimes a person can carry ESBL and not be sick but can spread ESBL to others. But because the person is not sick, no treatment is needed. ESBL infections in the elderly or compromised patients can be fatal if not treated properly.
VRE
Vancomycin is a powerful and often last-resort antibiotic. It is administered intravenously and must be closely monitored for therapeutic levels. Too little drug cannot kill the organism and can actually select for organisms that are resistant to the drug and too much can have harmful side effects, most often to the kidneys. Patient with some kidney failure are particularly sensitive to the drugs’ harmful effects. One of the organisms commonly treated with vancomycin is Enterococcus. Over time, this organism has evolved to resist vancomycin—thus the term Vancomycin Resistant Enterococcus. Patients being treated with vancomycin often have their blood levels of the drug checked at peak and trough. The peak level is the highest blood level obtained shortly after dosing of the patient, while the trough level is the lowest blood level measured immediately prior to the next dose. By measuring both the peak and trough, an effective drug level can be obtained without harmful side effects.
Mechanism of Drug Resistance Transfer
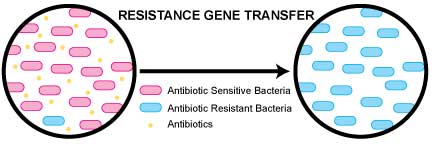
The reason that new resistances are occurring is because the gene that codes for the resistance is on an extra piece of DNA that resides outside of the bacteria’s genome. That separate piece of DNA can enter a new bacteria species and transfer the resistance to the new organism—allowing it to become resistant to the particular antibiotic.
Figure 1 shows how a mixed infection with two bacteria—one resistant to the prescribed antibiotic and one sensitive to the prescribed antibiotic—can become a resistant mixed infection following a resistance gene transfer.
Reporting
Most long-term care facilities want immediate notification of one of these antibiotic resistant organisms, to enable effective treatment and infection control measures. MEDLAB includes these resistant organisms on our critical call list.
Last Resort Treatment
When all possible antibiotics have been exhausted and the infection still persists, the only alternative is surgery to remove the infected tissue. Obviously this is not always possible and may actually spread the infection to surrounding area. Such practices could be termed the beginning of the “post-antibiotic” era. While new antibiotics are in the pipeline, many of the most powerful have undesirable side effects and face years of safety and regulatory hurdles.
Prevention
The laboratory, first and foremost, must identify antibiotic resistant infection. Laboratories should notify facilities when organisms are identified that might put other patients at risk. Since definitive organism ID and antibiotic sensitivity usually takes days to report, it is not considered a critical or panic value that triggers a call. However, such arrangements can be made if necessary or desirable. The notification should trigger appropriate clinical and infection prevention measures.
Staff should be educated about preventing transmission of these organisms and how to properly care for patients with infected with these organisms.
Hand washing is the most effective tool for combating any of the superbugs. Hand washing between rooms or between patients, along with an alcohol-based hand rub has greatly reduced the incidence and spread of such infections.
The other preventative measure is to prescribe the correct antibiotic for the particular organism known as an ID and Sensitivity test (or Culture and Sensitivity (CandS) or Minimal Inhibitory Concentration (MIC). The ID correctly identifies the organism, while the sensitivity tests the isolated bacteria against a variety of antibiotics. The “bug and drug” profile identifies which antibiotic is most effective for that particular patient’s organism. This practice is often referred to as antimicrobial stewardship.
Reducing the use of devices such as central venous catheters, endotracheal tubes, or urinary catheters that are known to be potential sources of device–associated infections is an important part of the effort to decrease the incidence of these infections.
It is up to each individual facility to make their own infection control guidelines, but most facilities house patients colonized or infected with any of the superbugs in single patient rooms and if not available, these patients should be cohorted together.
In summary, superbugs are prevalent in long-term care settings. Since the resistance gene can jump from one strain of bacteria to another—this problem is going to continue to grow. Couple this with the limited repertoire of available antibiotics, this is an early warning that effective identification, treatment and prevention should be undertaken to prevent infection and spread to the most vulnerable residents and patients in long term facilities.
Gary L. Milburn, Ph.D is the chief technical officer for MEDLAB.





