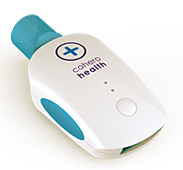
Cohero Health has received clearance from the U.S. Food and Drug Administration for its mobile spirometer.
The spirometer is wireless and hand held, and connects to a smartphone or tablet for real-time measurements of critical lung function metrics, the company said. It works with Cohero Health’s mobile asthma and COPD disease management platform, which uses Bluetooth sensors and mobile apps AsthmaHero and BreatheSmart to log and provide feedback on results.
Cohero Health is the first company to use an integrated connected health platform to offer both lung function measurement and medication.
Cohero Health is based in New York City and focuses on digital health.




